Overview:Treatments of Non-Alcoholic Fatty Liver Disease & Type 2 Diabetes
LBS-009, developed by Belite Bio™, a subsidiary company of Lin BioScience, is a first-in-class anti-RBP4 oral therapy targeting liver disease and type 2 diabetes, two age-related metabolic diseases. Non-alcoholic fatty liver disease/non-alcoholic steatohepatitis and type cumulatively impact more than 1.89 billion patients worldwide. Many studies in the peer-reviewed literature have focused on the role of RBP4 in both diseases, confirming that elevated levels of RBP4 are a contributing factor. The combined projected global market for NAFLD/NASH and diabetes is $79 billion dollars.
LBS-009 is currently in preclinical development.
Disease Profile:
Non-Alcoholic Fatty Liver Disease
Non-alcoholic fatty liver disease (NAFLD) occurs when an excess accumulation of fat damages the liver. Currently, it’s estimated that 1.46 billion patients suffer from NAFLD worldwide.
Over time, the liver damage and the associated inflammation can lead to the development of non-alcoholic steatohepatitis (NASH), which impacts more than 9 million patients in the United States alone. As the disease progresses, it can lead to cirrhosis and eventually, complete liver failure.
Currently, there are no approved treatments for NAFLD / NASH, and patients often have no symptoms in the early stages
Type 2 Diabetes
Type 2 diabetes (T2D) is a chronic disease that occurs when the body cannot effectively use insulin, the hormone that regulates blood sugar levels. The health impact of T2D is profound, potentially causing damage to the heart, blood vessels, eyes, kidneys, and nerves (Source: WHO). Type 2 diabetes is on the rise, with approximately 422 million patients globally (Source: WHO).
How it Works (Mechanism of Action):
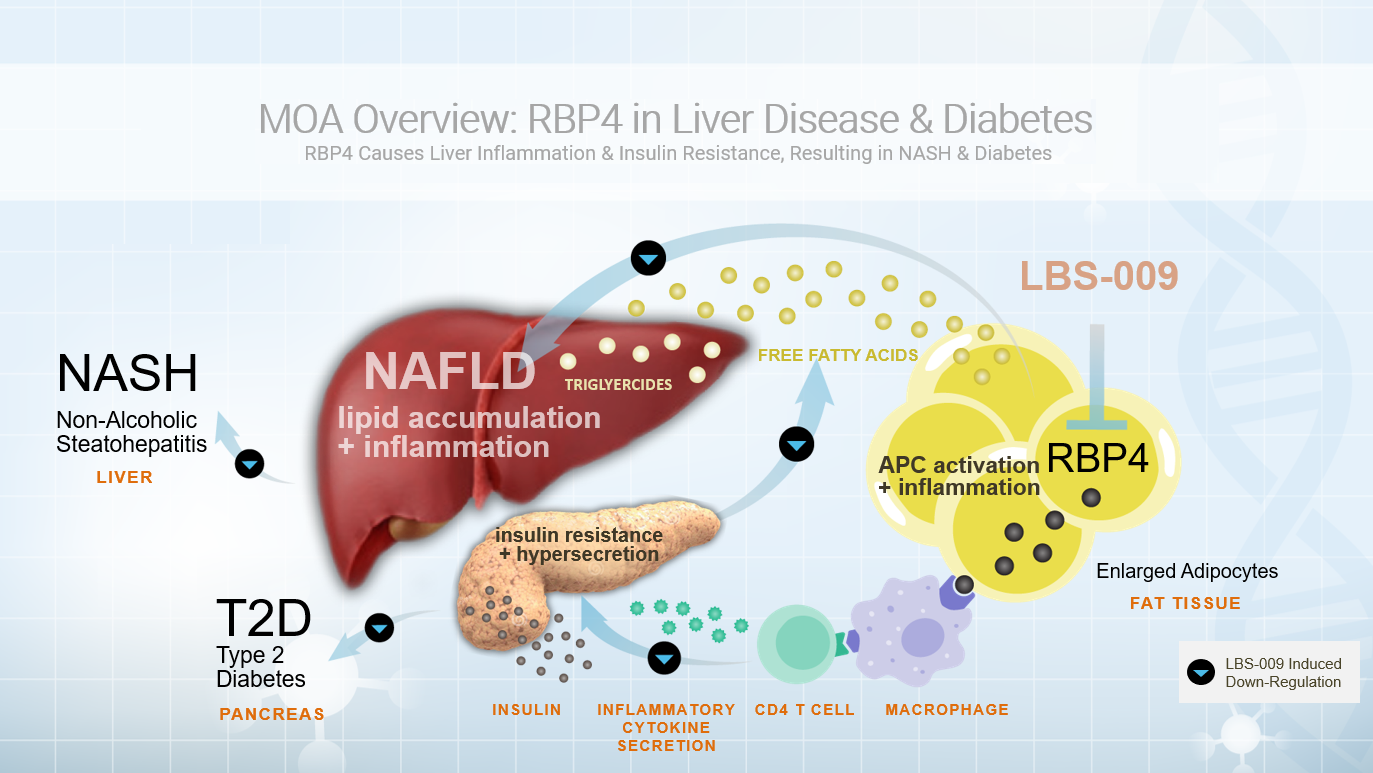
RBP4 has been identified as a pro-inflammatory cytokine in preclinical models of insulin resistance and liver disease and is currently being investigated as a clinical biomarker for human metabolic diseases. In response to the impaired glucose uptake associated with obesity, fat cells will increase their production and release of RBP4. Elevated concentrations of RBP4 in adipose tissue have been shown to activate antigen presenting cells (APC), resulting in an influx of immune cells.
This polarization and proliferation of CD4 T cells further amplifies inflammation, primarily in visceral adipose tissue, and is currently thought to be the driving force in RBP4-mediated insulin resistance. Increased RBP4 production in enlarged adipocytes also instigates a cascade of responses that result in a redistribution of free fatty acids from adipose tissue to the liver, leading to excessive accumulation of hepatic fat. While the role of RBP4 in inducing metabolic dysregulation is both complex and multifaceted, we believe modulating RBP4 concentrations systemically with LBS-009 has significant therapeutic potential for treating patients suffering from metabolically associated diseases.