Overview:First-in-Class Oral Therapy for Treatment of Dry Age-Related Macular Degeneration and Stargardt Disease
LBS-008, or Tinlarebant, if approved, would provide a novel treatment option where there currently is none. LBS-008 is an oral once-a-day treatment that can reduce and maintain the delivery of vitamin A (retinol) to the eye as a means to reduce the accumulation of toxic vitamin A by-products in ocular tissue. This effect is achieved through the ability of LBS-008 to reduce and maintain the level of serum retinol binding protein 4, or RBP4, which carries retinol from the liver to the eye. In clinical trials, LBS-008 has demonstrated its target specificity and potency that we believe could be clinically meaningful to treat Stargardt (STGD1) patients.
LBS-008 is a potent small molecule RBP4 antagonist that has been specifically designed to reduce the delivery of retinol to the eye as a therapeutic approach towards reducing the accumulation of cytotoxic bisretinoids, preserving the integrity of retinal tissues, and ultimately slowing or preventing loss of vision. The delivery of retinol to the RPE requires RBP4 and the RPE expresses a specific RBP4 receptor (STRA6) to regulate vitamin A uptake. Other extrahepatic tissues do not require delivery of retinol bound to RBP4 and do not express the RBP4 receptor. These tissues are able to take up vitamin A bound to non-specific carriers such as lipoproteins, triglycerides, and albumin.
LBS-008 is the only drug candidate within the current drug development projects of the National Institute of Health (NIH) Blueprint Neurotherapeutics Network, or the BPN, that is intended to treat dry AMD. The BPN was launched in 2004 to foster small-molecule neurotherapeutic development, bringing together a unique blend of grant dollars, industry-standard scientific expertise, and contract resources under a milestone-driven cooperative agreement program.
Additionally, the mechanism of action utilized by LBS-008 has been recognized and recommended as a priority for clinical development in both STGD1 and dry AMD in a systematic review published by the U.K. National Institute for Health Research, or the NIHR, in 2018. The NIHR screened 7,948 articles in 2018 for its systematic review on treatments for dry AMD and STGD1. Its principal findings included that research focus should be at earlier stages in both diseases (before vision is impaired) and that the most promising treatments for both diseases appear to be prevention of lipofuscin and bisretinoid accumulation. Therefore, the NIHR recommended the mechanism of RBP4 inhibition, which is utilized by LBS-008, as a promising treatment in dry AMD and STGD1.
The Phase 3 trial, DRAGON, to evaluate the safety and efficacy of LBS-008 in adolescent STGD1 patients has been commenced. Approximately 60 patients are targeted for enrollment in this study. For more information, visit clinicaltrials.gov at https://www.clinicaltrials.gov/ct2/show/NCT05244304?term=belite+bio&draw=2&rank=1. And a phase 2 or phase 3 clinical trial to evaluate safety and efficacy of LBS-008 in dry AMD is planned for initiation in 2022. LBS-008 has been granted fast track designation, rare pediatric disease designation (RPD) in the U.S., and orphan drug designation (ODD) in the U.S. and Europe.
Disease Profile:
Dry Age-Related Macular Degeneration (Dry AMD)
Age-related Macular Degeneration (AMD), or AMD, is a common eye disorder among people over 50. The only approved therapies for AMD are for ‘wet’ AMD, which represents approximately 10% of all AMD cases. There are no approved therapies for the other stages of AMD, including GA, secondary to advanced AMD, which are collectively referred to as dry AMD and represents approximately 90% of all AMD cases. There are an estimated 11 million dry AMD patients in the United States and over 196 million patients worldwide with an estimated global direct healthcare cost of US$255 billion.
Stargardt Disease
Stargardt disease (STGD1) is an inherited juvenile form of macular degeneration and currently, there is no approved treatment available. The disease is caused by a mutation in the ABCA4 gene, which leads to the accelerated formation and accumulation of toxic vitamin A by-products known as bisretinoids. The most prominent bisretinoid identified in human tissues is known as A2E (N-retinylidene-N-retinylethanolamine). The accumulation of A2E in ocular tissues causes progressive retinal cell death and permanent loss of vision. More than 500 mutations in the ABCA4 gene have been identified in STGD1 patients. Some STGD1 patients suffer severe visual impairment by the age of 20. The prevalence rate of STGD1 is estimated to be 1 in 10,000 people. Based on this estimate, approximately 30,000 US citizens are affected by STGD1. This estimate includes both adults and children.
.png)
Source: https://makariwellness.com/stargardt-disease/
How it Works (Mechanism of Action):
Preventing the Accumulation of Toxic A2E
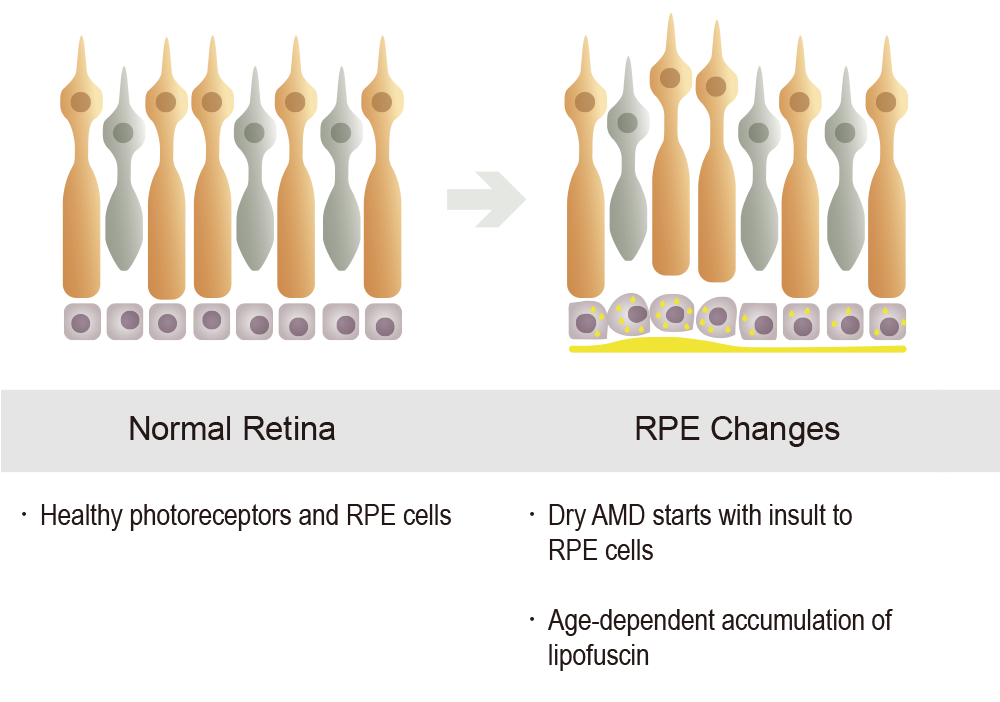
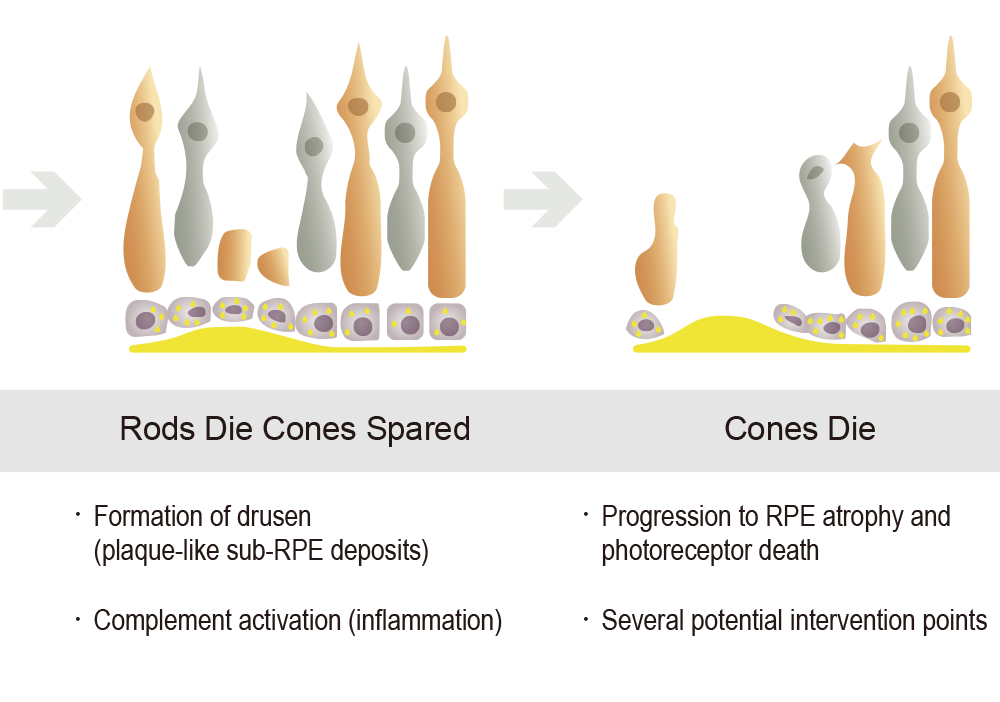
Both STGD1 and dry AMD are characterized by the early aberrant accumulation of lipofuscin and cytotoxic bisretinoids. The most abundant autofluorescent bisretinoid that has been identified in human lipofuscin is known as A2E (N-retinylidene-N-retinylethanolamine), a spontaneously formed complex comprised of two molecules of retinal and one molecule of ethanolamine. Investigations of the potential toxicity of A2E in cell-based assays and animal models have shown that this compound is highly toxic and can kill RPE cells in a concentration-dependent manner through myriad mechanisms. Because A2E and related bisretinoids are derived from vitamin A (i.e., they are by-products of normal visual cycle function), therapeutic approaches have focused on reducing levels of vitamin A (retinol) in the eye.
Visual Cycle
The processing of vitamin A (aka, retinol, or all-trans retinol) in the visual cycle begins with the delivery of circulating retinol to the RPE. The ternary complex of RBP4-retinol-TTR is presented to RBP4 receptors which are located on the basal surface of RPE. The RBP4-TTR vehicle serves to solubilize retinol and produce a large molecular size complex which resists elimination in the kidney. Upon entry into the RPE, retinol undergoes a series of enzymatic reactions resulting in generation of the visual chromophore, 11-cis retinal. The visual chromophore is delivered to the retina where it combines with opsin to form the light-sensitive visual pigment, rhodopsin. Photoactivation of rhodopsin liberates all-trans retinal which is transported out of the retina by the ABCA4 protein.
.png)